
To review:
The chemical structures that are involved in the ionization of acetic acid in water.
Introduction:
The water is a polar and universal solvent. Most of the molecules having ionic bonds dissolve in water by making bonds with it. The compound dissociates into the individual ions making the ionic bond weaker than the solid form of the compound.
Explanation of Solution
The acetoacetic acid dissociates into the acetoacetate
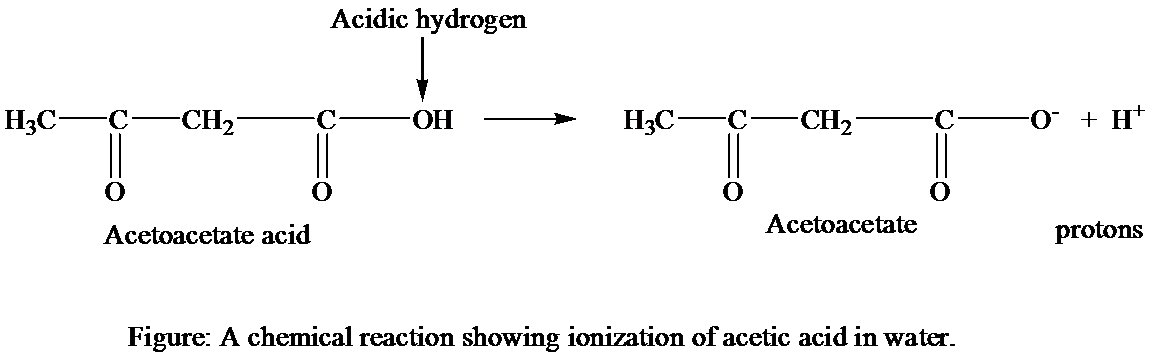
Thus, it can be concluded that the acetoacetate and hydrogen ions are chemical structures that are involved in the ionization of acetoacetic acid in water. The carboxyl group of acetoacetate infers the negative charge on the molecule and hydrogen ion gets dissociated from acetoacetic acid with its positive charge.
Want to see more full solutions like this?
Chapter 2 Solutions
Life: The Science of Biology