
a)
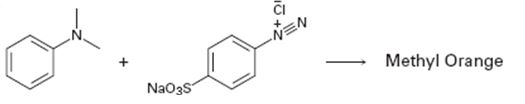
Interpretation:
A structure for methyl orange, an azo dye, produced when the two reactants shown react is to be drawn and the electron pushing mechanism for its formation is to be shown.
Concept introduction:
The diazonium cation can act as an electrophile and can attack
To draw:
The structure of methyl orange, an azo dye, produced when the two reactants shown react and to show the electron pushing mechanism for its formation.
b)
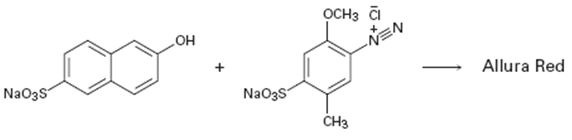
Interpretation:
A structure for allura red, an azo dye, produced when the two reactants shown react is to be drawn and the electron pushing mechanism for its formation is to be shown.
Concept introduction:
The diazonium cation can act as an electrophile and attack the aromatic rings. The dimethylamino group is an o- and p- directing group. Hence the diazonium cation can attack the ring at the p-position to yield a carbocation intermediate. The intermediate then can lose a proton to yield the desired product. This reaction is known as coupling reaction.
To draw:
The structure of allura red, an azo dye, produced when the two reactants shown react and to give the electron pushing mechanism for its formation.
c)
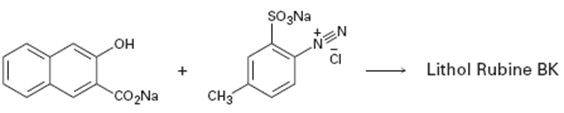
Interpretation:
A structure for lithol rubine BX, an azo dye, produced when the two reactants shown react is to be drawn and the electron pushing mechanism for its formation is to be shown.
Concept introduction:
The diazonium cation can act as an electrophile and attack the aromatic rings. The dimethylamino group is an o- and p- directing group. Hence the diazonium cation can attack the ring at the p-position to yield a carbocation intermediate. The intermediate then can lose a proton to yield the desired product. This reaction is known as coupling reaction.
To draw:
The structure of lithol rubine BX, an azo dye, produced when the two reactants shown react and to show the electron pushing mechanism for its formation.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- Write the appropriate reagents, conditions and products for the following electrophilic aromatic substitutions: COOH H₂ (3) Pt NO₂ ~arrow_forwardGive the expected major product of the following reaction: OH 1) LIAIH,arrow_forwardTriethanolamine, (HOCH2CH2)3N, is a widely used biological buffer, with maximum buffering capacity at pH 7.8. Propose a synthesis of this compound from ethylene oxide and ammonia.arrow_forward
- The reaction of N−bromosuccinimide with 4−methyl−3−nitroanisole has been reported in the chemical literature. This reaction yields a single product in 95% yield. Identify the product formed from this starting material.arrow_forwardThe following questions concern ethyl (2-oxocyclohexane)carboxylate.(a) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by a Dieckmann cyclization.(b) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by acylation of a ketone.(c) Write structural formulas for the two most stable enol forms of ethyl (2-oxocyclohexane)carboxylate.(d) Write the three most stable resonance contributors to the most stable enolate derived from ethyl (2-oxocyclohexane)carboxylate.(e) Show how you could use ethyl (2-oxocyclohexane)carboxylate to prepare 2-methylcyclohexanone.(f) Give the structure of the product formed on treatment of ethyl (2-oxocyclohexane)-carboxylate with acrolein (H2C=CHCH=O) in ethanol in the presence of sodium ethoxidearrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forward
- One frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forwardKetones and aldehydes react with sodium acetylide (the sodium salt of acetylene) to give alcohols, as shown in the following example: R1 ОН 1. НС—с: Na* R2 R1 R2 2. Нзо" HC Draw the structure of the major reaction product when the following compound reacts with sodium acetylide, assuming that the reaction takes preferentially from the Si face of the carbonyl group. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • You do not have to explicitly draw H atoms. • If a group is achiral, do not use wedged or hashed bonds on it. CH3 CHarrow_forwardBenzene + nitration, followed by bromination, then reduction of the nitro group to an amine, followed by diazonization, addition of copper I cyanide to the reaction and the product is O benzoyl nitrile O ortho-bromoaniline O para-bromo-nitrobenzene O 3,5-dibromnobenzonitrile O para-bromobenzyl bromidearrow_forward
- Indicate the products A and B that are obtained in the following reactions: OH CH3 + HOCH2-CH₂OH + TSOH → A 1° NaH/THF A + →> B 2º C6H5-CH2Cl Briefly comment on each reaction.arrow_forwardPlease provide a step by step list explaining the mechanism for the reactionsarrow_forwardDescribe how 3-methyl-1-phenyl-3-pentanol can be prepared from benzene. You can use any inorganic reagents and solvents, and any organic reagents provided they contain no more than two carbons.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



