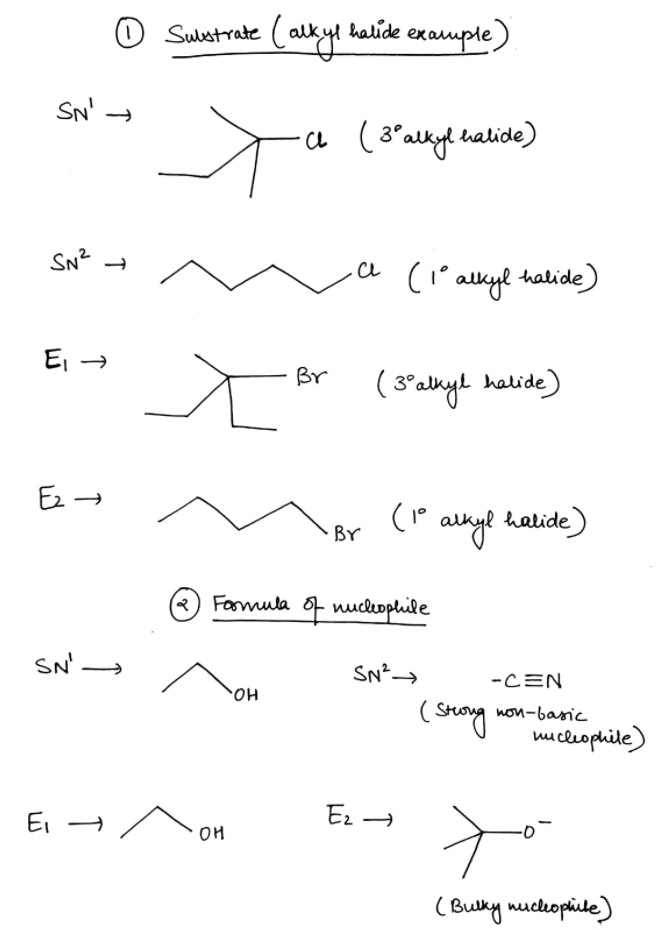
Fill in the information based on the type of reaction mentioned in each column. A. Draw the line angle diagram of a substrate that will undergo SN1, SN2, E1, E2 B. Draw the line angle diagram of a nucleophile that can be used for SN1, SN2, E1, and E2 C. Draw the line angle diagram of a solvent that can be used with SN1, SN2, E1, and E2 D. Write down if the stereochemistry of the product will be inversion, retention, or a 50:50 ratio of inversion and retention
Fill in the information based on the type of reaction mentioned in each column. A. Draw the line angle diagram of a substrate that will undergo SN1, SN2, E1, E2 B. Draw the line angle diagram of a nucleophile that can be used for SN1, SN2, E1, and E2 C. Draw the line angle diagram of a solvent that can be used with SN1, SN2, E1, and E2 D. Write down if the stereochemistry of the product will be inversion, retention, or a 50:50 ratio of inversion and retention
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter4: Recrystallization
Section: Chapter Questions
Problem 1Q
Related questions
Question
Fill in the information based on the type of reaction mentioned in each column.
A. Draw the line angle diagram of a substrate that will undergo SN1, SN2, E1, E2
B. Draw the line angle diagram of a nucleophile that can be used for SN1, SN2, E1, and E2
C. Draw the line angle diagram of a solvent that can be used with SN1, SN2, E1, and E2
D. Write down if the stereochemistry of the product will be inversion, retention, or a 50:50 ratio of inversion and retention

Transcribed Image Text:SN1
SN2
E1
E2
Substrate (alkyl halide
example)
Formula of
nucleophile
Formula of Solvent
Streochemistry of
product – retention or
inversion or both
Expert Solution
Step 1
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co